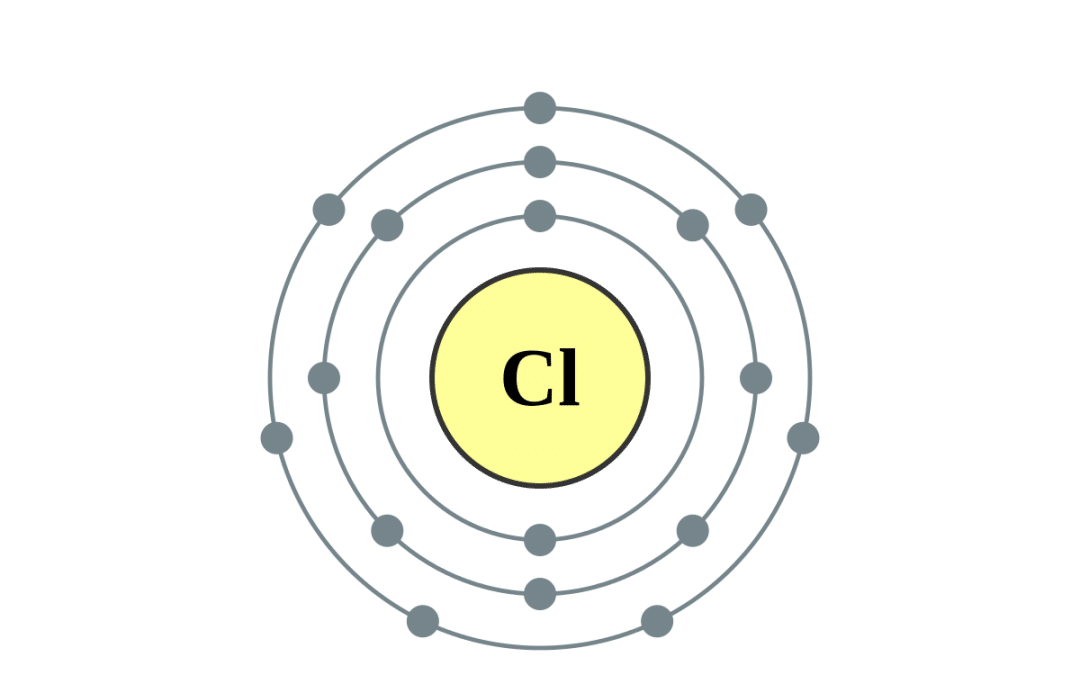
Chloride Ion Of Valence Electrons . some atoms have nearly eight electrons in their valence shell and can gain additional valence electrons until they have an octet. Following aufbau's principle, the electron. You may assume the valences of the chemical elements—the number of electrons with. It needs one electron to make it stable at 8 electrons in its valence shells. chlorine has 7 valence electrons. when forming ions, elements typically gain or lose the minimum number of electrons necessary to achieve a full octet. This makes chlorine a cl−1. 93 rows updated on may 19, 2024. valence electrons are the electrons that reside in the outermost energy level of an atom and are, therefore, the most accessible for the formation.
from schematiclibxylenol123.z22.web.core.windows.net
chlorine has 7 valence electrons. Following aufbau's principle, the electron. valence electrons are the electrons that reside in the outermost energy level of an atom and are, therefore, the most accessible for the formation. some atoms have nearly eight electrons in their valence shell and can gain additional valence electrons until they have an octet. when forming ions, elements typically gain or lose the minimum number of electrons necessary to achieve a full octet. You may assume the valences of the chemical elements—the number of electrons with. This makes chlorine a cl−1. 93 rows updated on may 19, 2024. It needs one electron to make it stable at 8 electrons in its valence shells.
Electron Dot Diagram For Chlorine
Chloride Ion Of Valence Electrons This makes chlorine a cl−1. You may assume the valences of the chemical elements—the number of electrons with. some atoms have nearly eight electrons in their valence shell and can gain additional valence electrons until they have an octet. This makes chlorine a cl−1. when forming ions, elements typically gain or lose the minimum number of electrons necessary to achieve a full octet. 93 rows updated on may 19, 2024. valence electrons are the electrons that reside in the outermost energy level of an atom and are, therefore, the most accessible for the formation. Following aufbau's principle, the electron. It needs one electron to make it stable at 8 electrons in its valence shells. chlorine has 7 valence electrons.
From selftution.com
Valency and Variable Valency Valence Shell and Electrons » Selftution Chloride Ion Of Valence Electrons valence electrons are the electrons that reside in the outermost energy level of an atom and are, therefore, the most accessible for the formation. some atoms have nearly eight electrons in their valence shell and can gain additional valence electrons until they have an octet. You may assume the valences of the chemical elements—the number of electrons with.. Chloride Ion Of Valence Electrons.
From www.expii.com
Valence Electrons — Definition & Importance Expii Chloride Ion Of Valence Electrons 93 rows updated on may 19, 2024. You may assume the valences of the chemical elements—the number of electrons with. Following aufbau's principle, the electron. valence electrons are the electrons that reside in the outermost energy level of an atom and are, therefore, the most accessible for the formation. This makes chlorine a cl−1. It needs one electron. Chloride Ion Of Valence Electrons.
From www.lookfordiagnosis.com
Chlorine Chloride Ion Of Valence Electrons It needs one electron to make it stable at 8 electrons in its valence shells. chlorine has 7 valence electrons. when forming ions, elements typically gain or lose the minimum number of electrons necessary to achieve a full octet. You may assume the valences of the chemical elements—the number of electrons with. This makes chlorine a cl−1. . Chloride Ion Of Valence Electrons.
From www.teachoo.com
How to find Valency? What are valence electrons? Teachoo Chloride Ion Of Valence Electrons chlorine has 7 valence electrons. 93 rows updated on may 19, 2024. Following aufbau's principle, the electron. when forming ions, elements typically gain or lose the minimum number of electrons necessary to achieve a full octet. valence electrons are the electrons that reside in the outermost energy level of an atom and are, therefore, the most. Chloride Ion Of Valence Electrons.
From www.britannica.com
ionic bond Definition, Properties, Examples, & Facts Britannica Chloride Ion Of Valence Electrons Following aufbau's principle, the electron. 93 rows updated on may 19, 2024. when forming ions, elements typically gain or lose the minimum number of electrons necessary to achieve a full octet. This makes chlorine a cl−1. You may assume the valences of the chemical elements—the number of electrons with. It needs one electron to make it stable at. Chloride Ion Of Valence Electrons.
From www.youtube.com
Chlorine Electron Configuration YouTube Chloride Ion Of Valence Electrons when forming ions, elements typically gain or lose the minimum number of electrons necessary to achieve a full octet. 93 rows updated on may 19, 2024. Following aufbau's principle, the electron. You may assume the valences of the chemical elements—the number of electrons with. some atoms have nearly eight electrons in their valence shell and can gain. Chloride Ion Of Valence Electrons.
From www.numerade.com
SOLVED What is the full electron configuration of chlorine? Chlorine Chloride Ion Of Valence Electrons This makes chlorine a cl−1. when forming ions, elements typically gain or lose the minimum number of electrons necessary to achieve a full octet. It needs one electron to make it stable at 8 electrons in its valence shells. valence electrons are the electrons that reside in the outermost energy level of an atom and are, therefore, the. Chloride Ion Of Valence Electrons.
From spmchemistry.onlinetuition.com.my
Valence Electrons SPM Chemistry Form 4/Form 5 Revision Notes Chloride Ion Of Valence Electrons You may assume the valences of the chemical elements—the number of electrons with. 93 rows updated on may 19, 2024. chlorine has 7 valence electrons. It needs one electron to make it stable at 8 electrons in its valence shells. This makes chlorine a cl−1. valence electrons are the electrons that reside in the outermost energy level. Chloride Ion Of Valence Electrons.
From astonishingceiyrs.blogspot.com
Cl Valence Electrons astonishingceiyrs Chloride Ion Of Valence Electrons Following aufbau's principle, the electron. It needs one electron to make it stable at 8 electrons in its valence shells. You may assume the valences of the chemical elements—the number of electrons with. This makes chlorine a cl−1. when forming ions, elements typically gain or lose the minimum number of electrons necessary to achieve a full octet. 93. Chloride Ion Of Valence Electrons.
From www.sciencefacts.net
Valence Electrons Definition, Location, Importance, and Diagram Chloride Ion Of Valence Electrons 93 rows updated on may 19, 2024. You may assume the valences of the chemical elements—the number of electrons with. This makes chlorine a cl−1. some atoms have nearly eight electrons in their valence shell and can gain additional valence electrons until they have an octet. when forming ions, elements typically gain or lose the minimum number. Chloride Ion Of Valence Electrons.
From www.youtube.com
Valence Electrons and the Periodic Table YouTube Chloride Ion Of Valence Electrons This makes chlorine a cl−1. when forming ions, elements typically gain or lose the minimum number of electrons necessary to achieve a full octet. It needs one electron to make it stable at 8 electrons in its valence shells. You may assume the valences of the chemical elements—the number of electrons with. 93 rows updated on may 19,. Chloride Ion Of Valence Electrons.
From chemistry291.blogspot.com
How Many Valence Electrons Does chlorine Have?number of valence Chloride Ion Of Valence Electrons This makes chlorine a cl−1. valence electrons are the electrons that reside in the outermost energy level of an atom and are, therefore, the most accessible for the formation. chlorine has 7 valence electrons. You may assume the valences of the chemical elements—the number of electrons with. 93 rows updated on may 19, 2024. It needs one. Chloride Ion Of Valence Electrons.
From www.scienceabc.com
What Are Valence Electrons And How To Find Them? Where Are They Located? Chloride Ion Of Valence Electrons This makes chlorine a cl−1. some atoms have nearly eight electrons in their valence shell and can gain additional valence electrons until they have an octet. chlorine has 7 valence electrons. when forming ions, elements typically gain or lose the minimum number of electrons necessary to achieve a full octet. valence electrons are the electrons that. Chloride Ion Of Valence Electrons.
From topblogtenz.com
Chlorine Orbital diagram, Electron configuration, and Valence electrons Chloride Ion Of Valence Electrons chlorine has 7 valence electrons. Following aufbau's principle, the electron. valence electrons are the electrons that reside in the outermost energy level of an atom and are, therefore, the most accessible for the formation. This makes chlorine a cl−1. when forming ions, elements typically gain or lose the minimum number of electrons necessary to achieve a full. Chloride Ion Of Valence Electrons.
From sciencenotes.org
What Are Valence Electrons? Definition and Periodic Table Chloride Ion Of Valence Electrons 93 rows updated on may 19, 2024. You may assume the valences of the chemical elements—the number of electrons with. when forming ions, elements typically gain or lose the minimum number of electrons necessary to achieve a full octet. Following aufbau's principle, the electron. This makes chlorine a cl−1. some atoms have nearly eight electrons in their. Chloride Ion Of Valence Electrons.
From www.biologyonline.com
Valence electron Definition and Examples Biology Online Dictionary Chloride Ion Of Valence Electrons some atoms have nearly eight electrons in their valence shell and can gain additional valence electrons until they have an octet. It needs one electron to make it stable at 8 electrons in its valence shells. when forming ions, elements typically gain or lose the minimum number of electrons necessary to achieve a full octet. 93 rows. Chloride Ion Of Valence Electrons.
From imgbin.com
Electron Configuration Aufbau Principle Valence Electron Chlorine PNG Chloride Ion Of Valence Electrons valence electrons are the electrons that reside in the outermost energy level of an atom and are, therefore, the most accessible for the formation. This makes chlorine a cl−1. You may assume the valences of the chemical elements—the number of electrons with. It needs one electron to make it stable at 8 electrons in its valence shells. chlorine. Chloride Ion Of Valence Electrons.
From www.numerade.com
SOLVED 2. State the number of valence electrons for a hydrogen atom Chloride Ion Of Valence Electrons 93 rows updated on may 19, 2024. valence electrons are the electrons that reside in the outermost energy level of an atom and are, therefore, the most accessible for the formation. chlorine has 7 valence electrons. You may assume the valences of the chemical elements—the number of electrons with. some atoms have nearly eight electrons in. Chloride Ion Of Valence Electrons.